
This material is well-known for its resistance to environmental factors like corrosion. This is a prime point for crevice corrosion.Stainless steel is an alloy made from iron, chromium, and other metals. Use of plastic washers, sealants and the like is useful because it prevents the formation of crevices, for example I fill my shroud swaged fittings with hot grease or Waxoyl to prevent entry of water. An active surface is only present when chemical action prevents the formation of the passive film, crevice corrosion being one of these cases. Stainless steels passivate in air within a very short period from being made. Polishing helps to remove these, so is a "good thing". Where staining occurs on shroud wires and similar components it is caused by very small cracks present in the surface, due to wire drawing with poor or insufficient lubricant. This accounts for staining commonly seen around small gaps, such as threads, under fittings, etc. Otherwise the normal carbon content is perfectly OK.ģ00 series alloys are good in general corrosion but are susceptible to crevice corrosion. 
These alloys are designated 304L and 316L. Both are non-magnetic.Ĭarbon content needs to be kept low for welded construction in corroding conditions. 316 stainless is the same composition plus the addition of 2% molybdenum for increased resitance to pitting in chlorides. 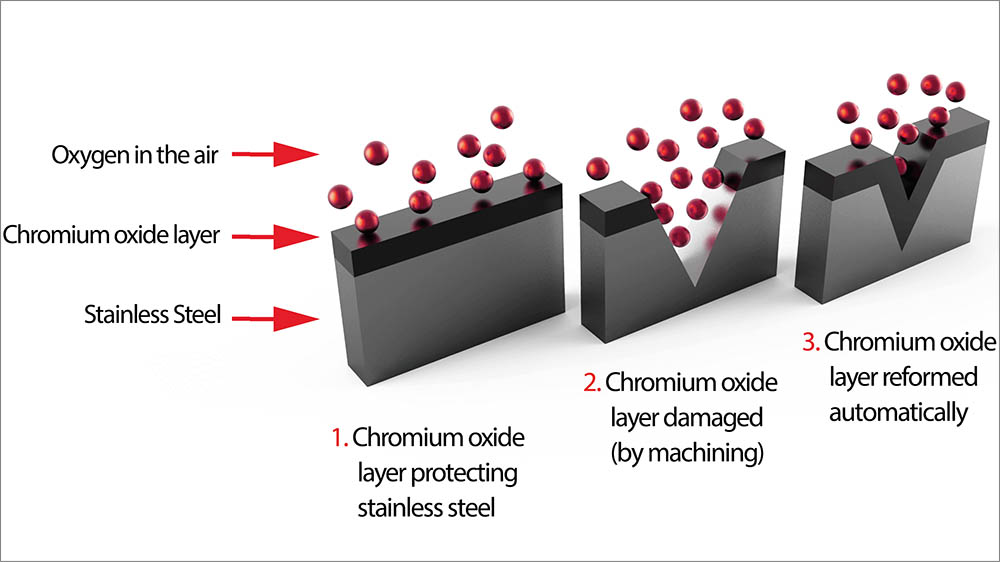
Its general corrosion resistance is provided by the chromium content. Its phase structure is austenite, referred to as austenitic. 304 stainless steel is 18 Cr, 8 nickel, balance iron. Not quite all about rusty stainless steel 316Ī few misconceptions here. Please respond if you have usefull additions and experiences Expect occassional small dings and bashes from tools to remove the passive layer: re-treat the area as above Light oils/wd40 etc will protect the general surfaceĤ. Diluted nitric acid will dissolve anyremaining carbon impurities and allow the passive layer to re-form.ģ. The surface layer of impurities must be heavily ground off, with a polishing/buffing wheel and paste, using a series of new polishing wheel covers and repeated washdowns, returning a high polish to the surface.Ģ. If you use a hackssaw to cut stainless steel, expect the sawn araea to be active, and remove steel deposits from the blade by grinding down, polishing as below.ġ. But any maintenance and aftercare must avoid use of carbon steel: removing rust from stainless steel should be with a plastic or fabric but NOT wire wool or mild steel brushes which will make rust return even faster/worse. Once passive, the parts should be greased up and isolated with plastic washers, clingfilm if out of signt, and so on.Ĥ) poor maintenance: of course, if a problem has not occurred as above, then maintenance should be minimal. If possible, two pieces of stainless steel should be isolated with plastic washers, but movemnt (eg under o-rings) can remove that passive layer.ģ) poor assembly: engineers have tendency to clean all parts before assembly - but should not do so with stainless steel. So stainless steel conectors/fasteners should always be tight, to avoid movement, and stainless steel chains or chained connectors may not resist rust for long in the presence of seawater. Big problem for any factory producing mild steel and stainless steel components.Ģ) fretting at the passive layer whilst in use anything that rubs on the stainless steel may remove that passive layer. drilling holes with high-carbon drills, cleaning with steel wool, using tools that have previously been working on mild steel, especially bending dies and so on. Secondly, it's a diferent material, so with an active 316 and a passive 316 touching each other, then together with seawater as electrolyte, there'll be galvanic corrossion - you'll see this as perhaps one stainless steel nut and bolt going rusty when everything else is fine.ġ) poor manufacturing- by introducing carbon impurities. Technically, 316 shouldn't rust - if it does, the passive layer has broken down.īreaking down the passive layer does two things :įirstly, the material is less resistant to rust: so the ends of a gurdaril wire covered in plastic rubs away at the s/s underneath, removes the passive layer and hence rusty ends of guardrail wires, rough/sawn stainless going rusty at the end, a light sprinkling of rust over a whole component, and so on. Once the surface layer has formed, the steel is known as 316(passive). They said "it's 316" and I said "ah but." and having explained to theme the rudiments of the problems, they're gong to sort it out.Įssentially, stainless steel is steel with additives, and these aditives are such that a layer of chroium oxide forms on the surface - hence "stainless". I'm having a moderate run-in with a new boat supplier about rusty stainless steel.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
